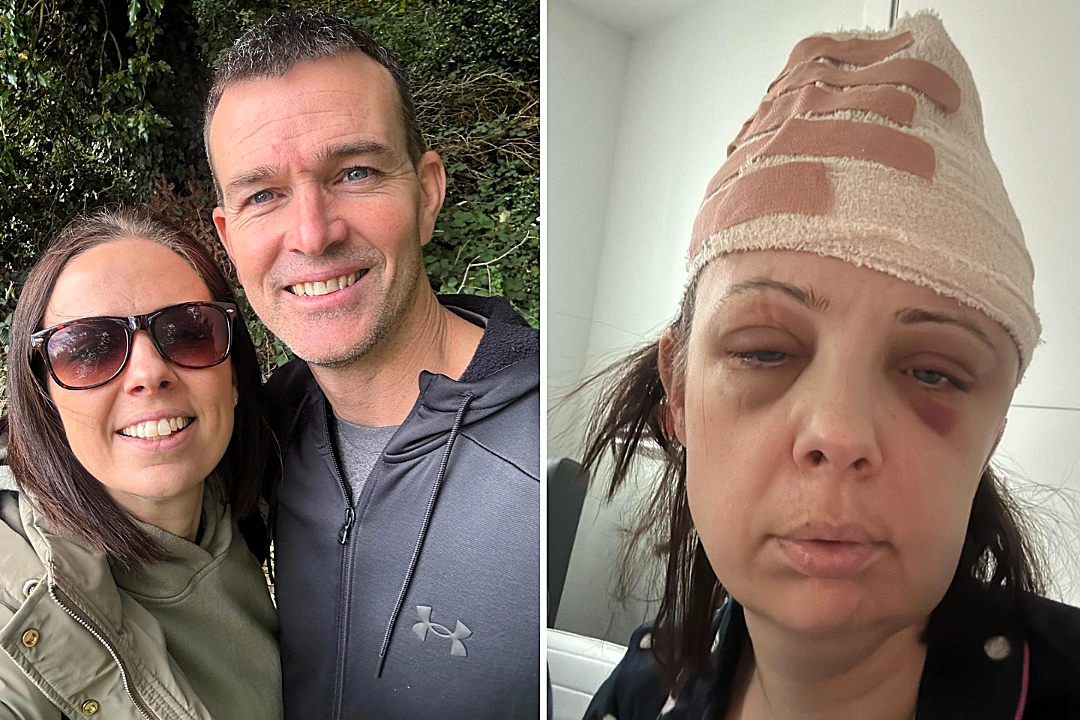
A mum who feared she would miss her children growing up after being diagnosed with a brain tumour says she has been given “her future back” thanks to a groundbreaking new drug.
Sally Penny was “terrified” when she suffered a seizure at home in and was rushed to hospital, where scans revealed she had an oligodendroglioma, a type of brain tumour.
The diagnosis left the 43-year-old fearing the worst.
READ MORE: British vet hospitalised after ‘dirty’ cat sinks teeth into her hand
Sally, who has two children, Fearne, 18, and Leo, 15, said she “cried solidly for about three months” as she struggled to come to terms with the life-changing news.
“I just couldn’t believe it,” Sally, from Hemel Hempstead, Herts, told Need To Know.

“I was shocked and terrified and so unbelievably sad because of all the things I thought I would miss in my children’s lives.
“I thought I wasn’t going to see them go to university, graduate, get married or get their first houses. I cried solidly for about three months.”
Determined to act quickly, she underwent surgery to remove the tumour.
Surgeons were able to remove 95% of it but the fear of what lay ahead remained.
That changed when she was offered access to a new drug, vorasidenib, through a special scheme.
Sally said: “I felt so lucky.
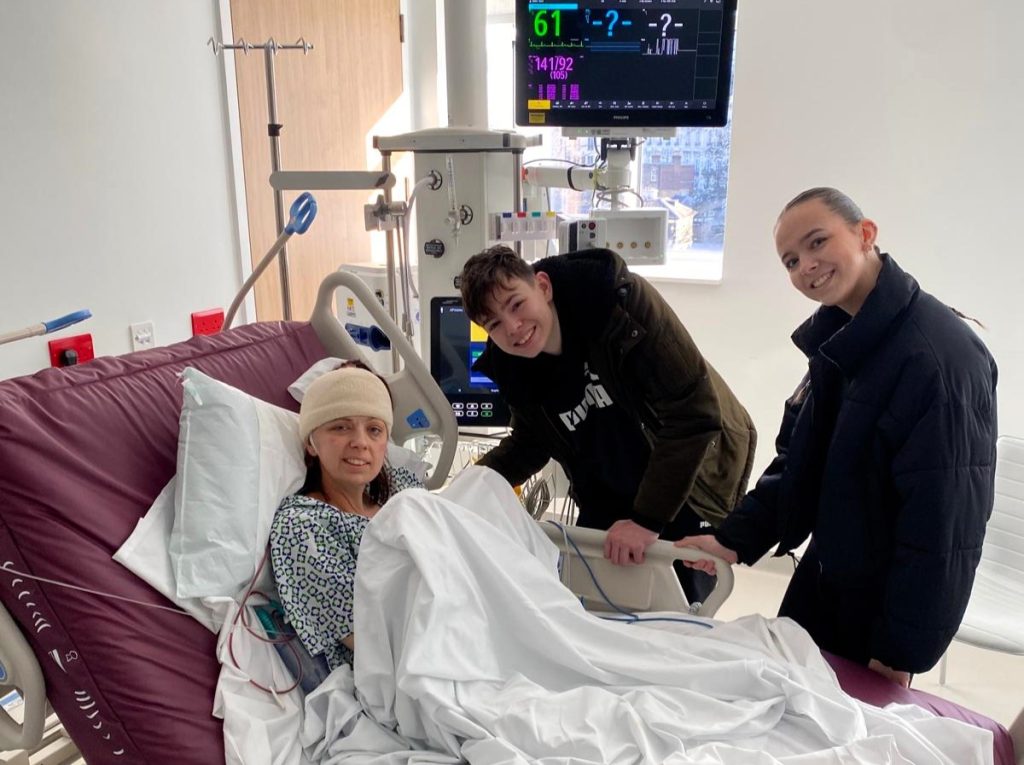
“When my husband and I went to collect the drug, we took loads of photos of the little pot of pills.
“We were so happy.
“When you have a brain tumour it’s quite hard to find little bits of hope, but this was hope and I felt like vorasidenib gave me a proper chance.
“I now take a daily tablet which has meant I haven’t needed to have radiotherapy or chemotherapy.”
Now, in what campaigners have described as a “historic moment”, the treatment has been approved for routine use on the NHS in England and Wales for eligible patients.
The drug will be offered to patients who have undergone surgery, but who are not in need of immediate radiotherapy and chemotherapy. It is the first targeted systemic therapy approved for IDH-mutant low-grade glioma.
For Sally, the impact has been life-changing.
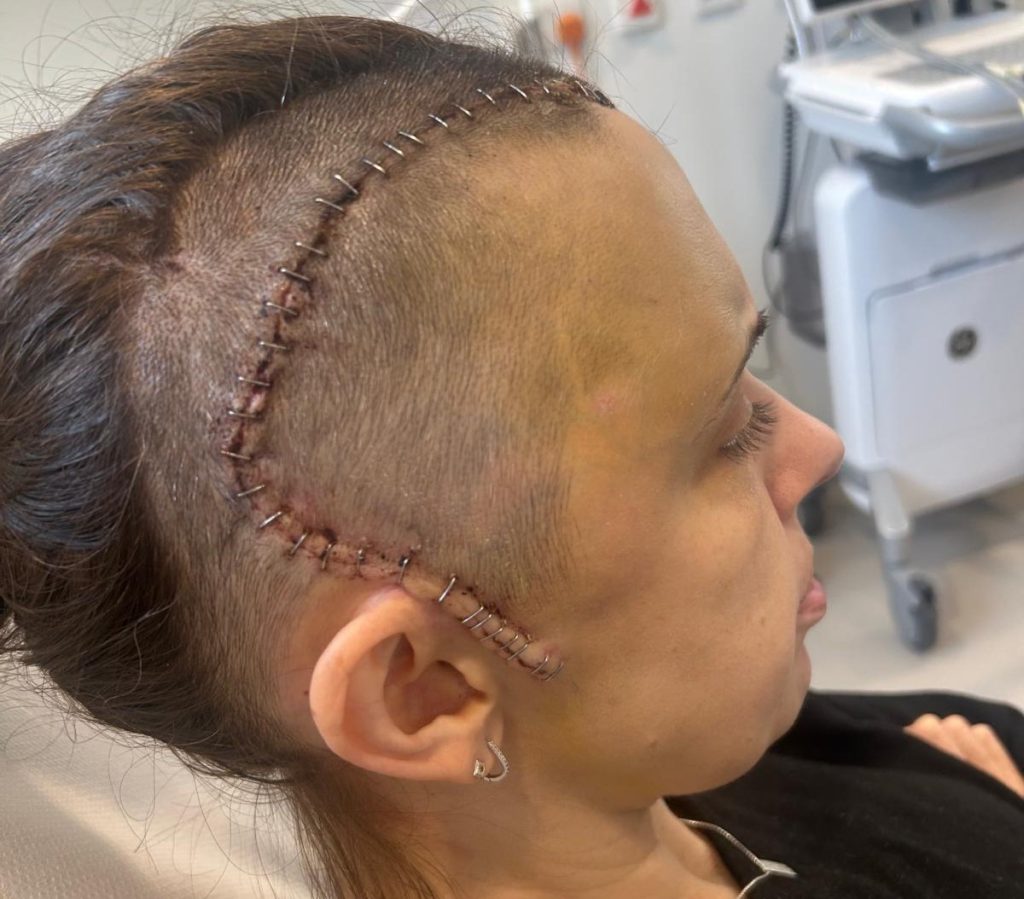
Sally Penny after surgery. (Jam Press/Brain Tumour Research)
She has been taking the daily tablet for a year and says four MRI scans have been stable.
Sally said: “Because of vorasidenib, I’ve got many years ahead of me so I will be able to witness the important milestones in my children’s lives which is fantastic.
“I have such confidence in the drug because I know a lot of other brain tumour patients on vorasidenib who have started to see some shrinkage of their tumours.
“I’m absolutely thrilled that vorasidenib has been approved by NICE for the NHS.
“I feel I can breathe a big sigh of relief as I’ll now be able to access the drug for as long as I need it and can continue my life feeling well and living as normally as possible by keeping my residual tumour stable.
“For patients like me who’ve been on the special access scheme, we can have continuity of a drug that’s working for us.
“But I’m just as excited for those who missed the scheme and will now be able to access this wonder drug and feel the sense of calm I feel going into each scan, knowing that vorasidenib has got my back.”
Dr Karen Noble, Director of Research, Policy and Innovation at Brain Tumour Research, said: “We are so pleased that patients with certain types of low-grade glioma will be able to access vorasidenib on the NHS in England and Wales.

Sally with her mother a few days after the operation. (Jam Press/Brain Tumour Research)
“This new treatment has been shown to significantly extend progression-free survival, meaning patients are able to go longer without needing radiotherapy or chemotherapy, and therefore without suffering the harsh side effects of these treatments.
“Brain Tumour Research has followed this approval process with keen interest and contributed to consultations.
“Our thanks to our community for sharing your stories to help support the case for this new drug.
“Vorasidenib is the first new treatment to be approved for adult brain tumours in the UK for more than 20 years.
“This is a hugely significant moment, but there is still a long way to go.
“We will continue to campaign for governments to invest more in research into brain tumours and to ensure that patients have access to life-changing clinical trials and new therapies.”
Brain tumours remain the biggest cancer killer of children and adults under 40, yet receive a fraction of national research funding compared to other cancers.
Brain Tumour Research funds sustainable research at dedicated centres in the UK.
It also campaigns for the Government and larger cancer charities to invest more in research into brain tumours in order to speed up new treatments for patients and, ultimately, to find a cure.
The charity is the driving force behind the call for a national annual spend of £35 million in order to improve survival rates and patient outcomes in line with other cancers such as breast cancer and leukaemia.